Author: Lee Benson MD, FRCPC, FACC, MSCAI, FPICS
How I do it: Tips, Tricks, and Techniques
A PICS Society education series
RVOT stenting in the symptomatic neonate.
Introduction:
Recent studies have observed that in the standard-risk symptomatic neonates with Tetralogy of Fallot (ToF), surgical repair beyond 3 months of age has perioperative and neurodevelopmental advantages. As such, where the decision is to perform a palliative procedure, a transcatheter intervention may be preferable. In the high-risk symptomatic neonate transcatheter palliative procedures is reasonable to consider as a risk management strategy. One such interventional procedure is stenting of the right ventricular outflow tract, which can be performed in ToF with PS or ToF with membranous valve atresia. [1] [2]
Anticipated challenges of the procedure:
- Maintaining adequate oxygenation (child may not have a patent arterial duct).
- Cannulation of the RVOT
- Angiographic definition of the infundibular, valve and MPA anatomy
- Secure placement of a coronary wire into a distal PA
- Choosing the correct length of the RVOT stent to cover the muscular outlet.
- Maneuvering the stent to the landing zone
TIP 1. Planning and Preparation
- Access: right femoral venous cannulation. Arterial access is optional.
- Imaging: Initially a RV angiogram in the cranial-LAO and lateral projections to outline the infundibulum-pulmonary valve-MPA and PA branches (Video 1). A second angiogram can be obtained in the outflow tract to better define the landing zone (Video 2). A post-implant angiogram in the outflow to assure complete coverage from the os infundibulum to the MPA.
- It is important to have the stent extend from the MPA (across the pulmonary valve) and completely cover the muscular outlet (os infundibulum), otherwise muscular obstruction will develop at the proximal (RV) end of the stent.
- As we are addressing the symptomatic neonate, there is no effort to avoid or spare the pulmonary valve (with a subvalvar implant) as in this setting, the pulmonary valve is significantly hypoplastic and would require a transannular patch.
Tip 2. Tools needed
- Sheaths: A 5 Fr. sheath for venous access. I use the Terumo Slender as the OD is only 1 Fr. size larger than the ID. If the child is very small (the smallest we have done is 1800 grams) it can be done with a 4 Fr. system.
- Catheters: For the RV angiogram: 5 Fr. Berman (Arrow) angiographic catheter. For the RVOT angiogram I use either a 4 or 5 Fr. RCA catheter with a 2.5 curve. The 4 Fr. RCA catheters we use (Cordis Inc.) have a slightly longer tip and are softer than the 5 Fr. RCA catheters (Cook Medical) which can be an advantage when cannulating the RVOT (Figure 1). Both have 0.035" lumens. Some operators will use a 5 Fr. coronary guide placed in the infundibulum to deliver the stent or place a Flexor sheath (Cook Medical) in the RA for extra backup for stent delivery.
Devices: Coronary BMS: Integrity (Medtronic Inc), Liberte (Boston Scientific) or Pro-Kinetic Energy (Biotronik) are in our stock, with no preference of one platform over the other. Some operators have used larger bare metal stents (JoStent, Abbot Vascular, Genesis, Cordis Corp, Formula 414 Cook) for children >3.5kg [3] - Other equipment: A Touhy-Borst adapter for contrast injection through the RCA catheter while having wire support. Insufflator for the coronary stent.
Tip 3. How I do it
Details of the technique:
Cardiac catheterization is carried out under general anaesthesia with endotracheal intubation and ventilation. Drugs prepared before the procedure include esmolol, and phenylephrine.
Access is from the femoral vein. Heparin 100 IU/kg is given to achieve an ACT >250sec. Biplane angiography of the right ventricle is performed to obtain an overview of the anatomy, with special attention to the 90° left lateral to identify the location of the os infundibulum and pulmonary valve plane, to make measurements. Most common tube angulation is 30° right anterior oblique (RAO) with 20° cranial tilt together with a 90° left lateral projection. (Video 1).
My preference is to perform an injection in the outflow tract with the RCA catheter, to better define the infundibular anatomy and make measurements of the length of the landing zone (as noted above) and chose the stent diameter. Generally, I will pick a diameter 1 to 2 mm larger than the MPA, regardless of the pulmonary valve annular dimension (Video 2).
A trick to avoid cannulating the aorta through the VSD, when placing the RCA catheter into the infundibulum is to rotate the catheter counter-clockwise. This will allow the catheter to move anteriorly, rather than toward the VSD. But you have to complete the rotation, to avoid getting stuck in the RV free wall.
A coronary wire (Wizdom Cordis) is then placed into the distal pulmonary artery. Generally, it goes easiest into the RPA, but either branch is fine. It is important to get it as distal as possible. I then guide the RCA catheter over the wire into the proximal RPA. A second coronary wire for better support (GrandSlam Asahi) is positioned into the RPA and the first wire removed. The guide wire distal soft end will often bend upon itself (Figure 2), allowing support from the wire shaft. A trick to improve wire support is to twist the wire in a clock-wise direction until the soft tip is looped in the lung parenchyma. This will fixate the wire and improve tractability of the stent. (Just remember which way to turned the wire so you can release the tip!).
Now with the wire in place, you are set to deploy the stent (Video 3). Remember that you must cover the os infundibulum past the pulmonary valve annulus to avoid acquired obstruction. If you are too short do not hesitate to telescope a second stent to complete the coverage (Videos 4, 5 and 6). If there is difficulty in maneuvering the stent into the infundibulum, despite the use of the back-up wire, and you decide not to fixate the wire distally, you can place a long sheath (such as a Flexor sheath) in the right atrium for added support or as mentioned some operators have placed an MP shaped guide catheter into the RV or an RCA guide into the outflow.
Although not detailed in this short summary of the procedure it is possible to consider stenting the outflow tract in the setting of membranous pulmonary atresia where the pulmonary valve can be perforated and stent positioned as outlined above.
Pitfalls to avoid
- Do not rely on incomplete angiography for measurements and landing zone assessment.
- Assure that the wire position is secure, and there is enough support to maneuver the stent.
- Do not hesitate to implant a second stent to fully cover the RV outflow.
- Avoid jailing one of the branch PAs with a too distal stent implant.
Tip 4. What complications to expect and how to deal with them
- If the RVOT stent is required for a prolonged implant time, in-stent stenosis can occur. As such, restenting is required (see Video 7)
- If the stent is misplaced, e.g., too proximal, consider tacking it down with a second stent. In this regard, it is critical not to lose wire position. Retrieval techniques used for misplaced conduit stents may be tried such as using a balloon to reposition the stent. Unfortunately, more often than not, the stent will be caught in the cords of the TV, and it would be more prudent to go to surgery. Fortunately, this can be avoided by good anatomical definition and stent placement.
- We have not encountered stent induced arrhythmias, although they have been reported, it is rare. Similarly, after deployment, TV regurgitation has not been a clinical issue in our experience.
- Arterial saturations in the presence of a patent arterial duct makes the conduct of the procedure simpler and where appropriate it is recommended that prostaglandins be administered. In the absence of a patent arterial duct arterial desaturation is not uncommon during manipulation in the right ventricular outflow tract. It is important to avoid hypotension and the administration of vasoconstrictor drugs such as phenylephrine is useful.
Summary:
RVOT stenting for the symptomatic neonate has proven to be an effective form of palliation to maintain adequate arterial saturations and promote pulmonary artery growth while awaiting elective surgical repair. The procedure requires a thorough understanding of the morphology of the outflow tract but from a technical standpoint relatively straight forward. Attention to anatomical details is paramount for success.
References:
- Quandt D, Ramchandani B, Penford G, Stickley J, Bhole V, Mehta C, Jones T, Barron DJ, Stumper O (2017) Right ventricular outflow tract stent versus BT shunt palliation in Tetralogy of Fallot. Heart 103: 1985-1991
- Miller JR, Stephens EH, Goldstone AB, Glatz AC, Kane L, Van Arsdell GS, Stellin G, Barron DJ, d'Udekem Y, Benson L, Quintessenza J, Ohye RG, Talwar S, Fremes SE, Emani SM, Eghtesady P (2023) The American Association for Thoracic Surgery (AATS) 2022 Expert Consensus Document: Management of infants and neonates with tetralogy of Fallot. J Thorac Cardiovasc Surg 165: 221-250
- Stumper O, Ramchandani B, Noonan P, Mehta C, Bhole V, Reinhardt Z, Dhillon R, Miller PA, de Giovanni JV (2013) Stenting of the right ventricular outflow tract. Heart 99: 1603-1608
Summary:
Figures with legends:
Figure 1

Note the slightly more gradual curve compared to the 5 Fr. RCA
Figure 2
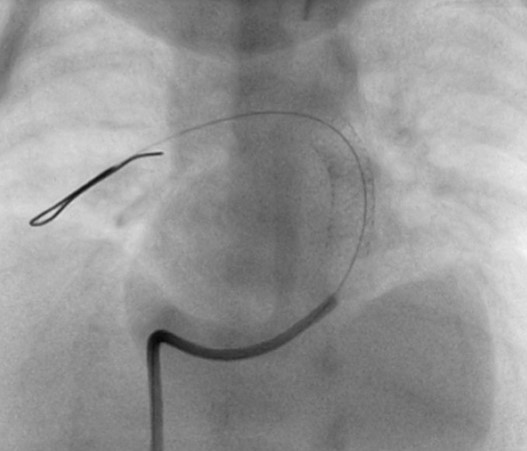
LAO-cranial. A coronary wire is placed in the distal RPA. Note that the soft distal end has folded into itself to allow more wire shaft in the PA for better support. The wire can also be rotated to embed it into lung tissue for additional support.
Video 1:
Panel A: LAO-cranial and B: 90° left lateral of the initial RV angiogram using the RCA catheter (by hand injection). While informative, the details of the outflow anatomy are best seen by a selective injection in the infundibulum.
Video 2:
Panel A: LAO-cranial and B: 90° left lateral of the selective injection into the infundibulum with the RCA catheter stabilized by a coronary wire in the RPA. Note the better anatomical definition for taking the measurements and identifying the potential landing zone.
Video 3:
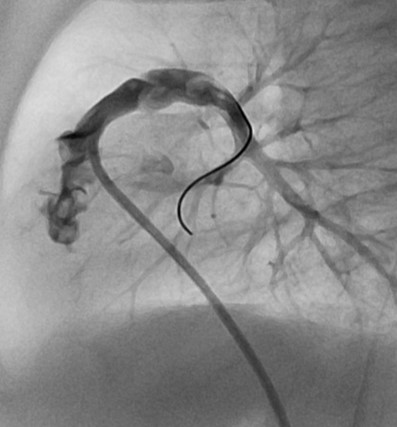
Panel A & B 90° left lateral.
In panel A, the stent is positioned in the RVOT using a reference still frame (panel B) from the selective Infundibular injection. Note the inflation is slow, such that small adjustments in positioning can be performed as the inflation begins.
Video 4:
Panel A LAO-Cranial and Panel B 90° left lateral. The stent has been implanted in the RVOT but has not covered the os infundibulum, resulting in persistent muscular obstruction. Distally, the stent is well opposed to the MPA, crosses the valve plane, but does not impinge on the PA confluence.
Video 5:
Video 6:
Panel A LAO-Cranial and panel B 90° left lateral. A second stent has been implanted, covering the full length of the outflow.
Video 7:
Panel A RAO-Cranial and Panel B 90° left lateral. In-stent stenosis has developed in the proximal aspect of the stent.
Panels C and E Slight-Cranial and Panel D and F left lateral. Positioning the stent to address the in-stent stenosis. Note in this procedure a 4 Fr. long sheath was used to provide extra-back-up for stent delivery.
Panel G Slight-Cranial and Panel H 90° left lateral. Final angiogram after stent placement.