Author: David Bloom, MD., David Balzer, MD
How I do it: Tips, Tricks, and Techniques
A PICS Society education series
Internal PA banding ("Flow Restrictors")
Introduction:
Pulmonary artery banding (PAB) is a technique used to control pulmonary blood flow in patients with complex single ventricle anatomy, or patients who will require complex biventricular repairs later in infancy to reduce surgical mortality. Although historically a surgical procedure, a transcatheter approach to internal PAB has been shown to be feasible. The first reports of transcatheter stage I palliation using a modified Amplatzer device were published in 2001 [1], however the concept of transcatheter PAB or pulmonary flow restrictors (PFR) continues to be an area of ongoing research. The use of modified Microvascular Plugs (MVP) (Medtronic Inc., Minneapolis, MN) was first reported in a swine model in 2019 [2]. This study demonstrated the feasibility of using the modified MVP for PFR. Importantly, the study demonstrated that this technique allowed for transcatheter retrieval of the device up to 9-12 weeks following implantation, without significant distortion of the pulmonary artery (PA) anatomy. There have been small cases series (Giessen, Germany, Washington University in St. Louis) [3, 4] reporting the use of a modified MVP for PFR with mixed results. With further development of technique and expansion of use, it is possible that transcatheter PFR using the widely available MVP will be a legitimate option for patients with single ventricle physiology, or high risk two ventricle patients with pulmonary overcirculation.
Anticipated challenges of the procedure:
- Device positioning: may jail upper branch of PA (although this is typically not an issue since the distal portion of the device is uncovered) or migrate distally in PA after device release therefore appropriate device sizing is critical.
- Retrieval: Snares should be available in case retrieval is necessary. Retrieval may be possible up to 12 weeks after the procedure [2]
- Thrombosis: Although this has not been an issue in our experience, the patient should be heparinized for the procedure and antiplatelet therapy should be considered following device deployment.
TIP 1. Planning and Preparation
- Access
- Imaging
- Multidisciplinary Discussion
Tip 2. Tools needed
- Sheaths
- Catheters
- The MVP 5Q, and 7Q can be delivered through a 4F catheter (recommended inner diameter (ID) of 0.027" and 0.041" respectively).
- The MVP 9Q will require a 5F catheter (recommended ID of 0.043").
- Devices
Tip 3. How I do it
- Details of the individual tip and technique
- Pitfalls to avoid
Tip 4. What complications to expect and how to deal with them
We have experienced device migration into the distal PA, as well as jailing of branches off of the PA, and this may require device retrieval and re-implantation acutely, or at a later time. Device retrieval is possible using an Amplatz Goose Neck Snare (Medtronic Inc, Minneapolis, MN) by capturing the proximal radiopaque marker on the MVP.
In our experience, despite attempts at creating a small hole in the modified MVP to limit pulmonary blood flow, some patients are still exposed to a significant left to right shunt, leading to pulmonary overcirculation and high mean PA pressure necessitating additional intervention (replacement of MVP) and/or medical management. This can be avoided by intraprocedural echocardiography to ensure that the PFRs are adequately restrictive prior to leaving the cath lab.
After removal of the MVP either transcatheter or at time of surgical intervention, it is possible that there will be stenosis and deformation of the branch PA anatomy (Figure 4) requiring catheter intervention and stenting.
Summary:
The use of transcatheter PFRs has the potential to allow for a totally percutaneous stage I approach in patients with single ventricle anatomy, and may allow for delaying surgical intervention in complex biventricular repairs in patients at high risk due to weight, age, or complex anatomy. There is limited data on the use of the modified Medtronic MVP, however it has proven to be a feasible quality option due to its accessibility and relative ease of use. Operators should be aware of the challenges and potential complications related to this procedure as described above. Ongoing research and development is necessary to further perfect the technique and devices used for this transcatheter procedure; additional studies comparing surgical PAB and transcatheter PFR are warranted as well.
References:
- Chan, K.C., C. Mashburn, and M.M. Boucek, Initial transcatheter palliation of hypoplastic left heart syndrome. Catheter Cardiovasc Interv, 2006. 68(5): p. 719-26.
- Khan, A.H., et al., Utility of the Medtronic microvascular plug as a transcatheter implantable and explantable pulmonary artery flow restrictor in a swine model. Catheter Cardiovasc Interv, 2019. 93(7): p. 1320-1328.
- Schranz, D., et al., Hypoplastic Left Heart Stage I: No Norwood, No Hybrid. Circulation, 2020. 142(14): p. 1402-1404.
- Nageotte, S., et al., Total Transcatheter Stage 1: A Word of Caution. Pediatr Cardiol, 2021. 42(6): p. 1410-1415.
figures
Figure 1
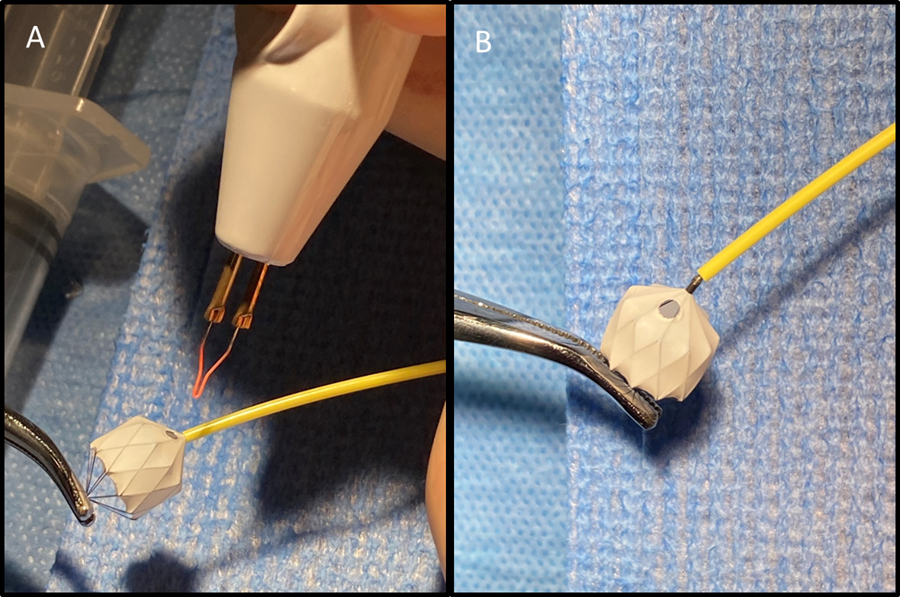
Figure 1. Modified Medtronic Microvascular Plug (MVP). A. Modification of one PTFE cell on inflow portion of device using low temperature fine tip Eye Bovie (Symmetry Surgical, Antioch, TN) cautery device to create 2-3mm hole. B. This example demonstrates the location and size of hole on MVP device.
Figure 2
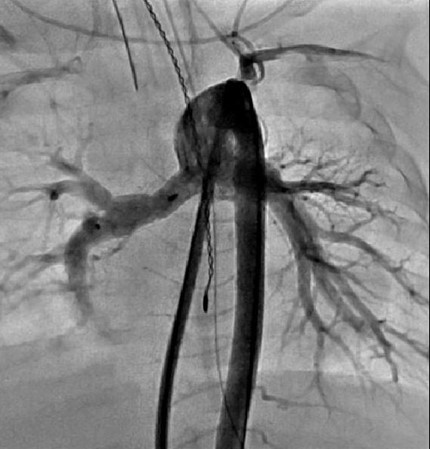
Figure 2. Angiographic evaluation of flow restrictors two weeks following placement. The PFR in the RPA jails the right upper pulmonary artery (RUPA), however this image demonstrates continued flow into the RUPA.
Figure 3
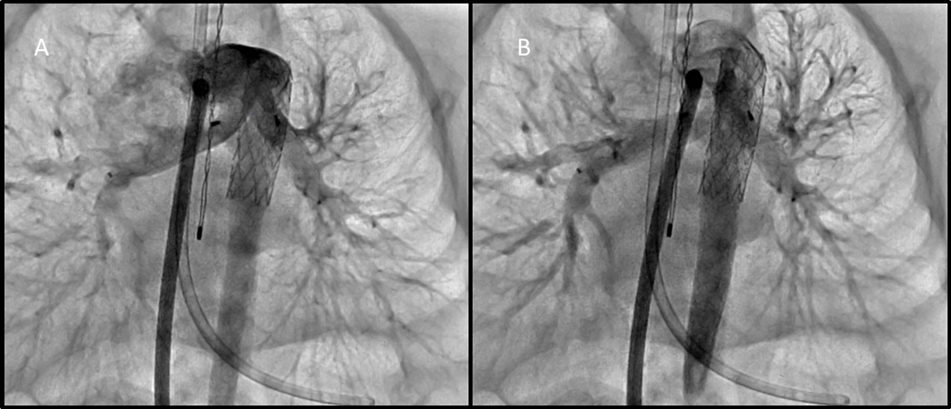
Figure 3. A. Angiographic evaluation of PFRs through a long sheath in the MPA. This first image better demonstrates jailing of the left upper pulmonary artery, with continued flow of contrast into this branch. B. This image demonstrates jailing of the right upper pulmonary artery. There is still flow seen entering this branch.
Figure 4
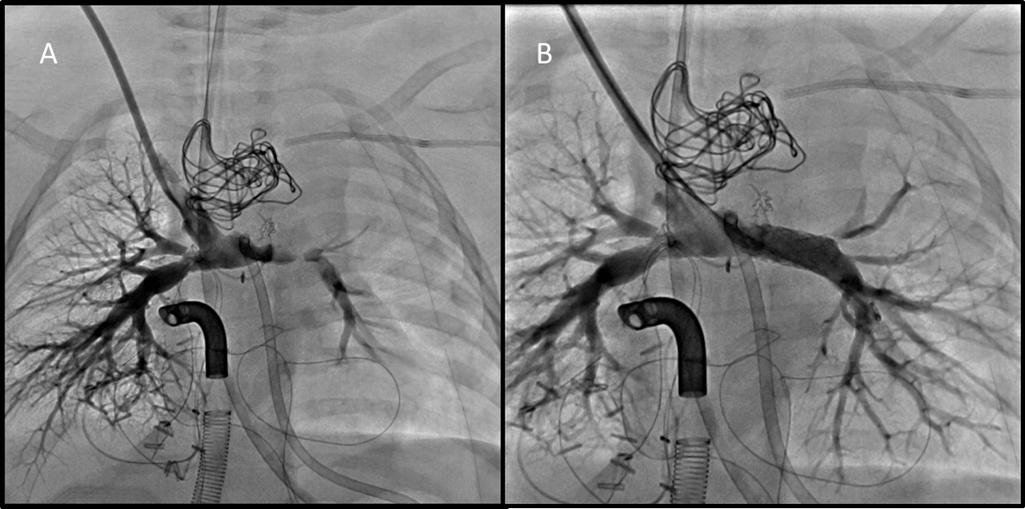
Figure 4. Angiographic evaluation of pulmonary arteries following comprehensive stage II palliation and surgical removal of PFRs. A. Angiogram in SVC demonstrates mild proximal RPA distortion, and moderate to severe LPA stenosis. B. Angiogram performed following 7mm stent placement in LPA showing significant angiographic improvement.